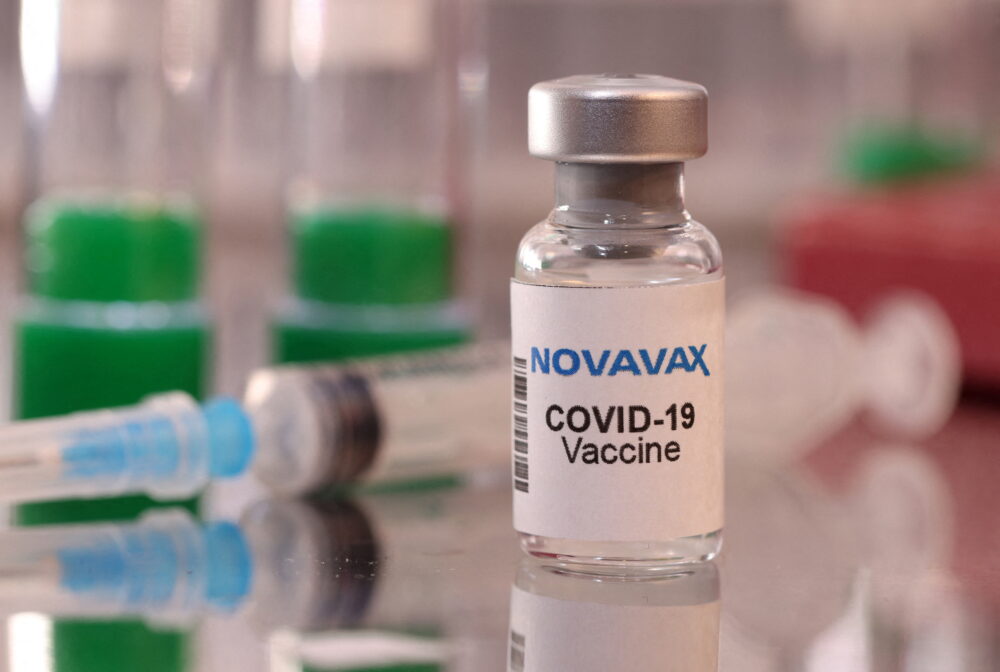
Novavax, a key player in the fight against COVID-19, has recently received authorization from the European Union for its updated COVID-19 vaccine. This development marks a significant milestone in the ongoing battle against the pandemic and showcases the company’s commitment to providing effective solutions for public health.
Key Highlights of the EU Authorization:
This authorization comes as global health authorities are continuously evaluating the effectiveness of vaccines against newly emerging variants. With the ongoing threat posed by COVID-19, having an updated vaccine that responds to these mutations is of paramount importance. Novavax’s latest vaccine release represents a proactive approach to this pressing issue, offering hope to many as nations prepare for potential new surges in cases.
Moreover, this approval could pave the way for further collaborations and innovations within the vaccine sector, strengthening international health measures against COVID-19. As more people receive the updated vaccine, the chance of widespread immunity increases, potentially leading us one step closer to returning to a sense of normalcy.
In conclusion, Novavax’s EU authorization for its updated COVID-19 vaccine is a hopeful turn in an ongoing public health campaign, emphasizing the vital role of vaccine advancement in managing and ultimately overcoming the impacts of COVID-19.








Leave a Reply